The Saudi Arabian Ministry of Health (MoH) has transitioned from a reactive oversight model to a proactive, credential-based regulatory framework for non-surgical cosmetic procedures. This shift addresses a critical failure in the previous market: information asymmetry between practitioners and patients. By mandating that Botox and dermal filler injections be performed exclusively by specialized physicians—dermatologists and plastic surgeons—the State is effectively raising the barriers to entry to eliminate "grey market" practitioners. This move is not merely about safety; it is a systematic restructuring of the health economy to centralize accountability and standardize clinical outcomes.
The Triad of Regulatory Risk Mitigation
The new mandates function through three distinct mechanisms that target the vulnerabilities of the aesthetic medicine supply chain.
1. Clinical Competency Constraints
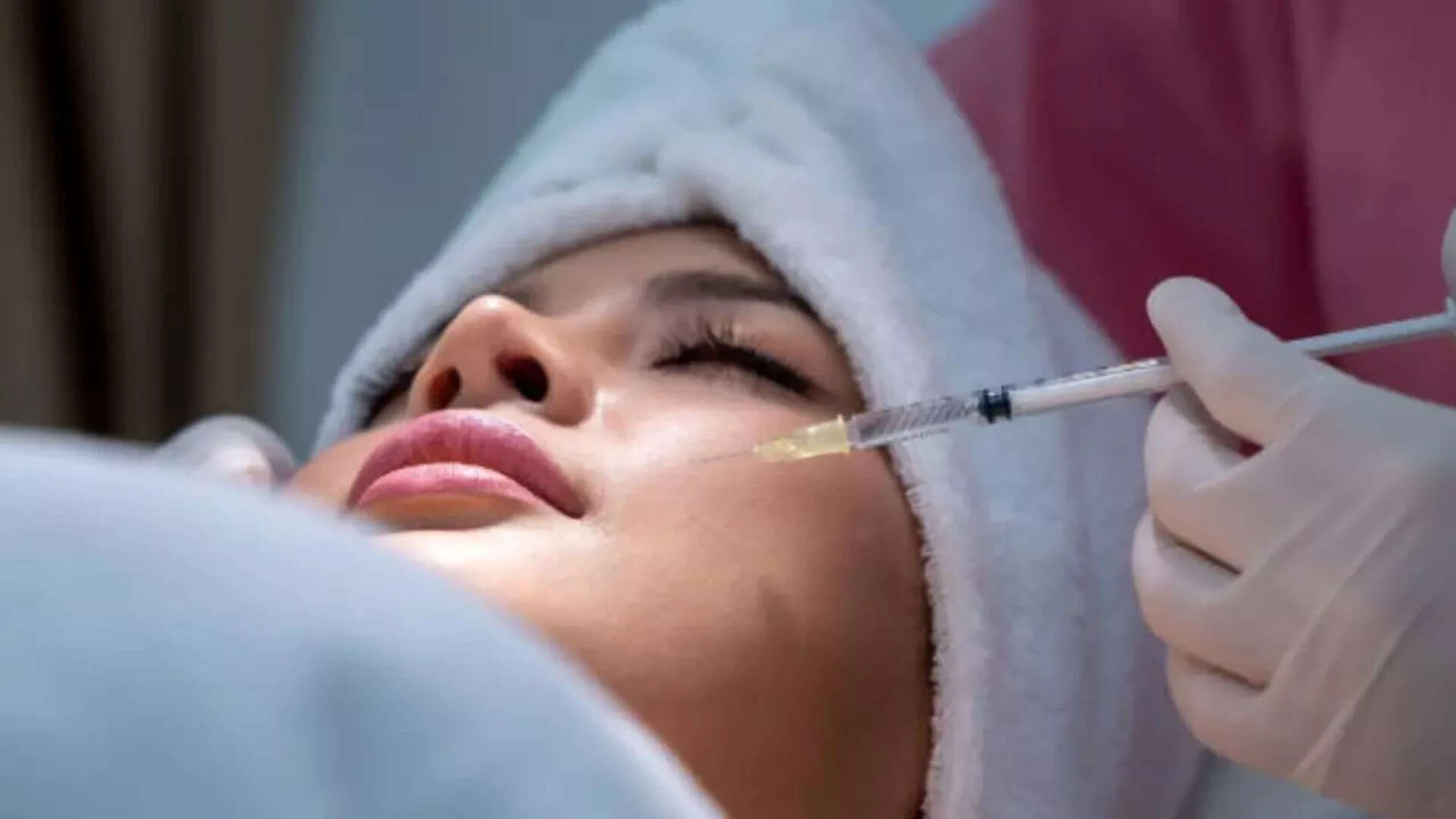
The core of the regulation lies in the restriction of practice to specific medical titles. Historically, the "aesthetician" or "general practitioner" could perform these injections under vague supervision. The MoH now recognizes that Botox (Botulinum Toxin) and fillers are not just cosmetic products but pharmacological agents with complex pharmacokinetic profiles.
Botox acts as a neuromuscular blocking agent. Its efficacy depends on precise anatomical placement within the musculature to avoid ptosis (drooping) or systemic diffusion. Fillers, conversely, are space-occupying implants. The primary risk here is vascular occlusion—the accidental injection of filler into an artery—which can lead to tissue necrosis or blindness. Only specialists with advanced training in facial anatomy and emergency reversal protocols (such as the use of hyaluronidase) possess the technical capacity to mitigate these high-stakes complications.
2. Supervision and Facility Accountability
By mandating "specialist supervision," the Ministry is closing the loophole of the "ghost clinic." In previous iterations of the market, a specialist might lend their name to a facility without being physically present during procedures. The current framework moves toward a model of direct liability. If a complication occurs, the supervising specialist is legally and professionally tethered to the outcome. This forces a shift in clinic operations:
- Operational Throughput: Clinics can no longer scale by hiring low-cost, mid-level providers to run parallel injection rooms.
- Labor Costs: The requirement for high-tier medical labor increases the "per-syringe" cost of operation, likely leading to a price floor in the legal market.
- Risk Premiums: Malpractice insurance and compliance costs will rise for facilities that previously operated with lean medical oversight.
3. Supply Chain Integrity
A secondary effect of tightening practitioner rules is the stabilization of the pharmaceutical supply chain. Authorized distributors of Botox (Allergan) and major filler brands (Restylane, Juvederm) typically only sell to licensed medical professionals with verifiable credentials. By restricting who can legally perform the procedure, the Ministry simplifies the detection of "cold chain" violations. Non-specialists who continue to operate must source products from the black market, where temperature control is rarely maintained, leading to protein denaturation in Botox or contamination in fillers.
The Economic Impact of High-Fidelity Standards
The Saudi aesthetic market is one of the fastest-growing in the MENA region. This regulation acts as a market filter. While it may temporarily reduce the volume of available providers, it increases the long-term "Life Time Value" (LTV) of the patient by reducing the rate of corrective procedures.
The Cost of Complication
From a systems-thinking perspective, unregulated aesthetics create a negative externality for the public health system. When a budget clinic botches a filler injection, the patient often ends up in an emergency room of a government hospital. The MoH is essentially internalizing these costs. By forcing the private sector to use expensive, highly-trained labor, the state reduces the downstream burden on public trauma and dermatology wards.
The Bifurcation of the Market
We are witnessing the emergence of two distinct tiers:
- The Compliant Tier: High-cost, high-safety clinics that use the MoH certification as a marketing tool. These entities will focus on "Natural Results" and "Medical Grade Safety."
- The Shadow Tier: Unregulated practitioners operating in residential spaces or salons. The Ministry’s crackdown includes heavy fines and permanent closure of such establishments, signaling that the cost of non-compliance is intended to be ruinous.
Technical Execution and Patient Safety Protocols
The MoH guidelines imply a standardized patient journey that practitioners must now document to remain compliant. This journey is broken down into four technical phases:
Pre-Procedure Diagnostic
A specialist must conduct a physical assessment of the patient’s skin elasticity, muscle strength (in the case of Botox), and existing volume deficits. This is the stage where contraindications—such as neuromuscular disorders or active infections—are identified. A general technician lacks the diagnostic depth to identify these "red flag" variables.
Product Verification
The regulation emphasizes the use of SFDA-approved (Saudi Food and Drug Authority) materials. Each vial or syringe has a trackable serial number. Under the new specialist-led model, the chain of custody for these substances is more easily audited during random ministry inspections.
The Injection Vector
The depth of injection determines the result. For example, Botox must be injected into the belly of the muscle, whereas different fillers require different planes (supraperiosteal vs. subdermal).
| Procedure | Injection Plane | Critical Risk |
|---|---|---|
| Botox | Intramuscular | Muscle atrophy or unintended paralysis |
| Hyaluronic Acid Filler | Subdermal / Deep Malar | Vascular compromise or granuloma |
| Biostimulators | Deep Dermis | Nodule formation |
Post-Procedural Monitoring
The mandate for specialist presence ensures that the "Golden Hour" after a complication is utilized. If a patient shows signs of blanching (a sign of vascular occlusion), a specialist can immediately administer the reversal agent. A non-specialist would likely delay treatment, leading to permanent scarring.
The Strategic Shift for Consumers and Investors
For the consumer, the "specialist only" rule serves as a heuristic for quality. The difficulty of navigating medical credentials is replaced by a simple binary: Is this person a certified dermatologist or plastic surgeon?
For investors in the Saudi healthcare space, the regulation suggests a consolidation phase. Independent, low-compliance salons will likely be phased out or acquired by larger medical groups that can afford the payroll of multiple specialists. Value is shifting away from "retail convenience" toward "clinical excellence."
The Ministry's move is a clear signal that the Saudi Vision 2030 goals for healthcare include the professionalization of the "wellness" sector. By treating beauty as a branch of medicine rather than a branch of service, the State is building a high-trust environment intended to attract medical tourism from neighboring Gulf states.
The immediate operational priority for any facility in the Kingdom is a full audit of practitioner licenses against the MoH "Specialist" database. Facilities must transition their marketing from "price-per-unit" to "clinician-led outcomes." Failure to re-align with this medical-first hierarchy will result in not just legal penalties, but total exclusion from the evolving Saudi healthcare ecosystem. The focus must remain on the surgical precision of the delivery model, treating the injection as a medical intervention with aesthetic benefits rather than a cosmetic service with medical risks.